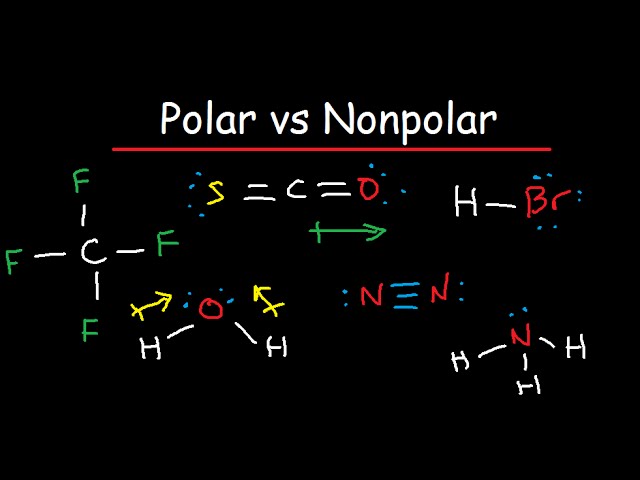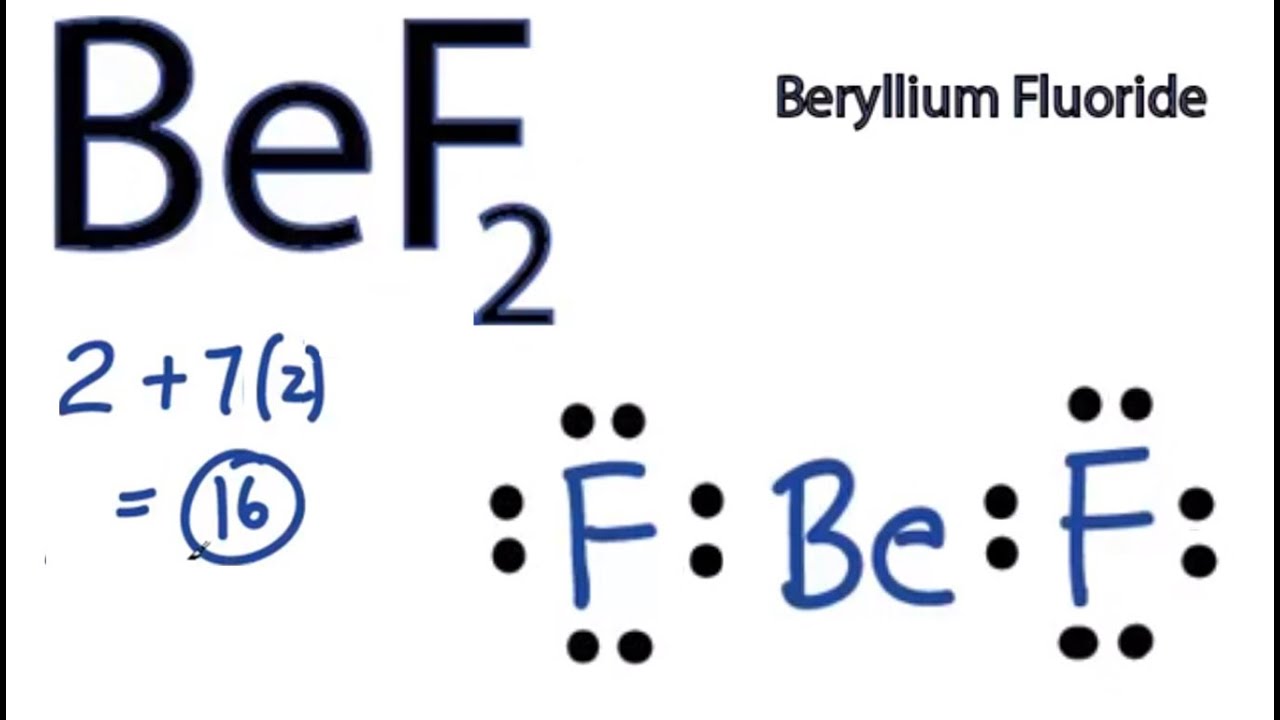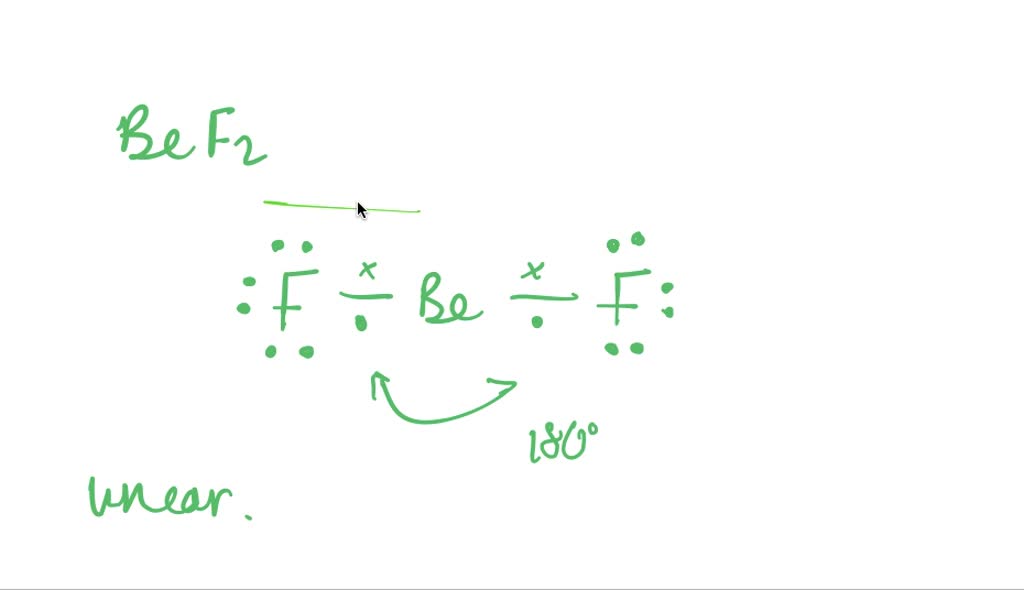
SOLVED:Despite the large electronegativity difference between fluorine and beryllium atoms, BeF2 forms linear molecules in gas phase. Are these molecules polar or nonpolar? Explain.

Is BeF2 and BF3 are nonpolar covalent compound as the vector sum is zero - Chemistry - - 10567947 | Meritnation.com
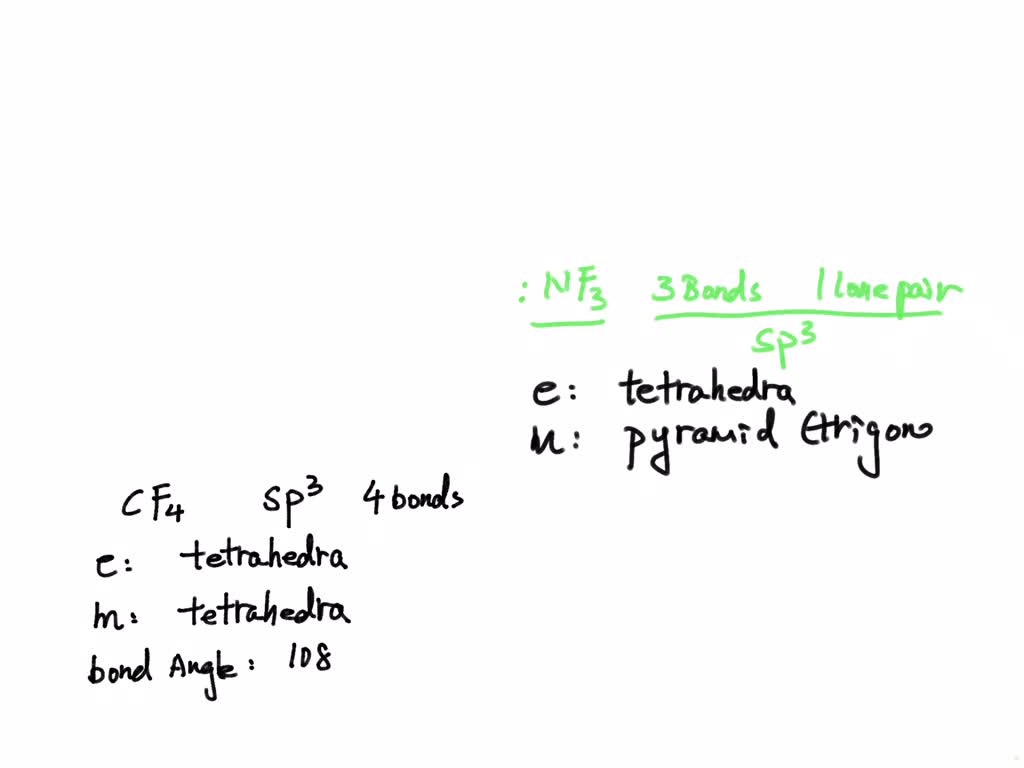
SOLVED: Molecule Geometry, Bond angle(s), polar or non-polar of CH2, HCN, HF, SO2, BeF2, SF5+, SF4, SF3-, BrF4-, BrF5, H2, O2, N2, and CH2Cl2