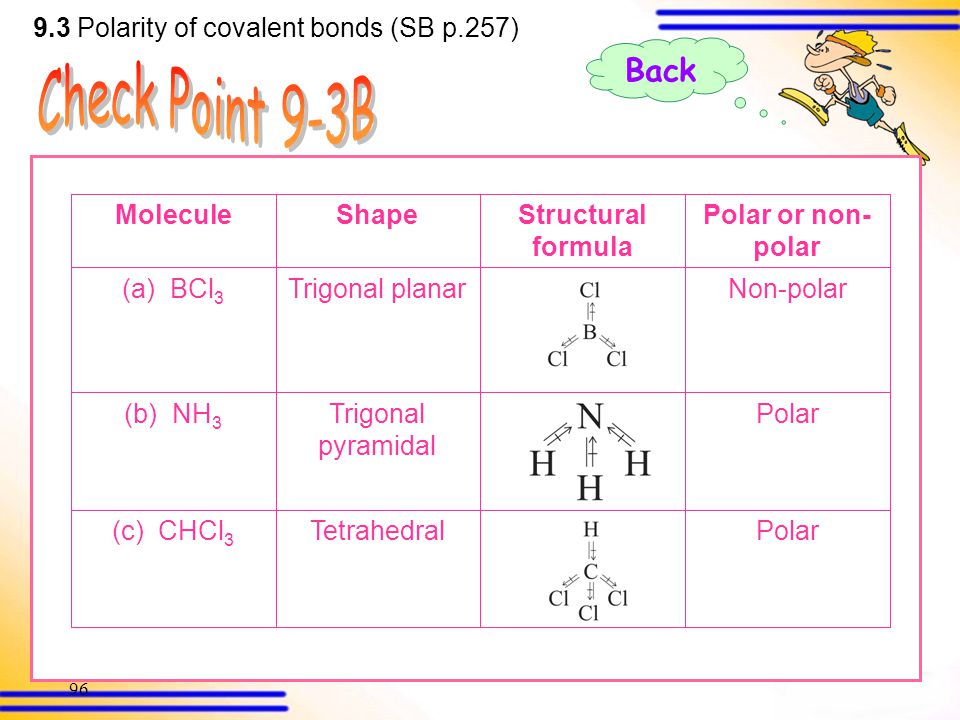
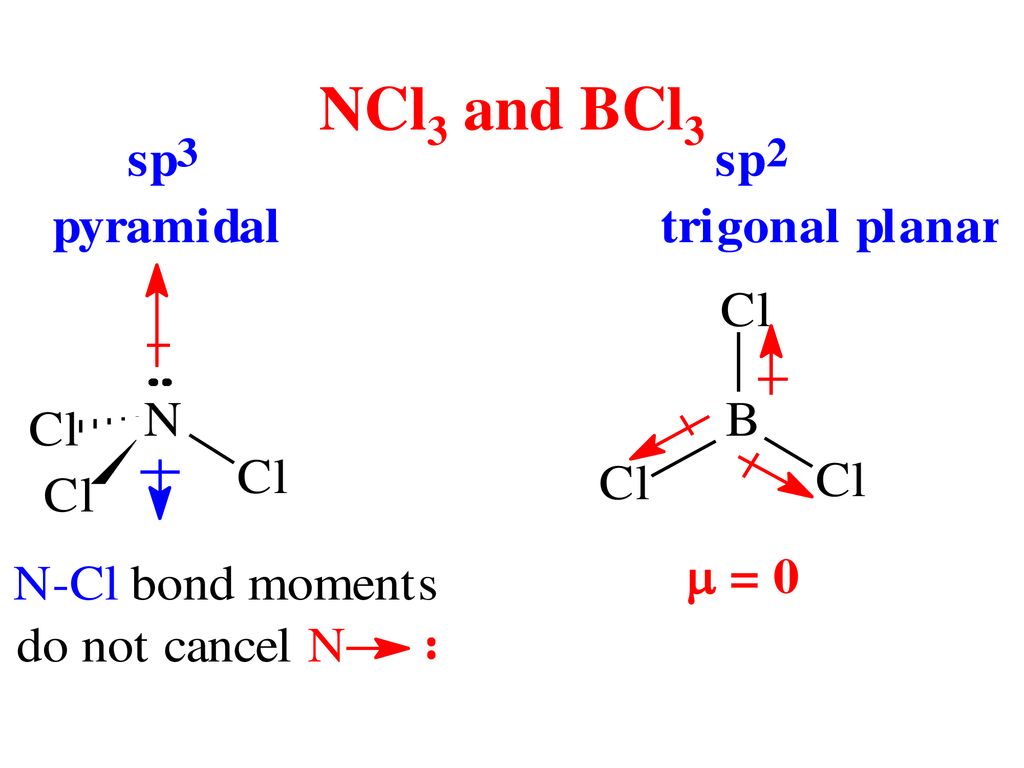
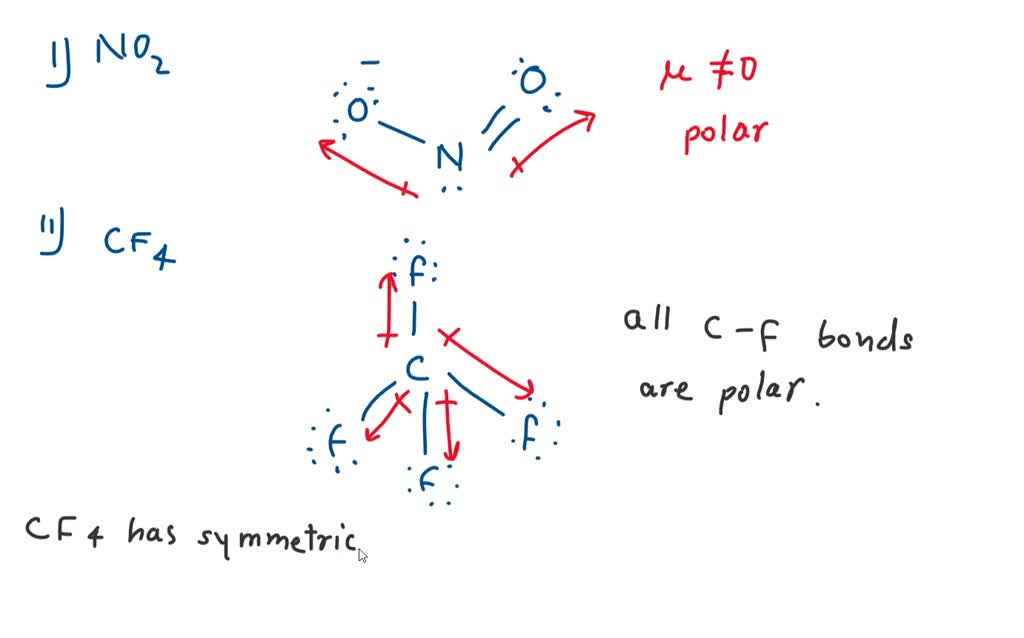
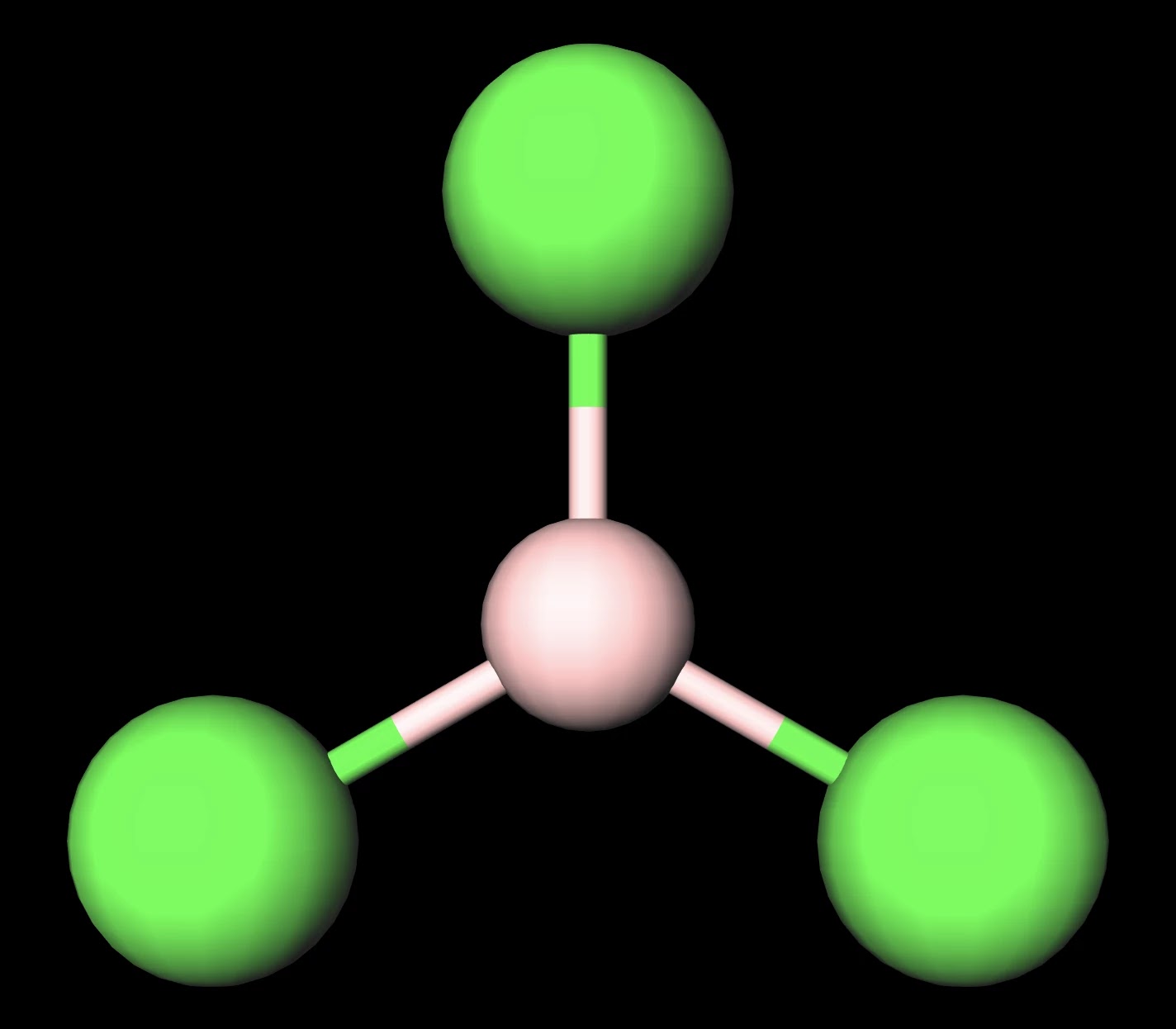
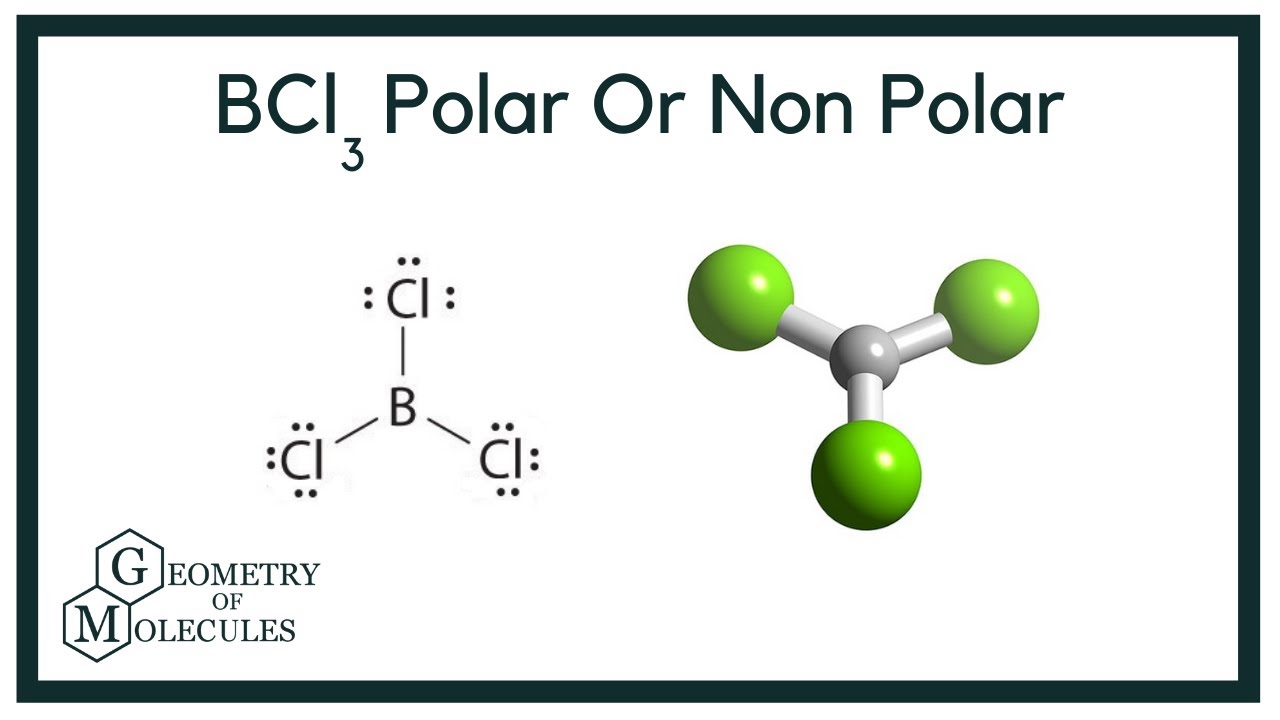
SOLVED: Molecular Structure: The VSEPR Model Examples: Momnemh Eq = Predict shape and polarity Y A. polar (u + 0) B. nonpolar (u = 0) PCl3 Vs BCl3 PCIs Vs SbCl;?- SF

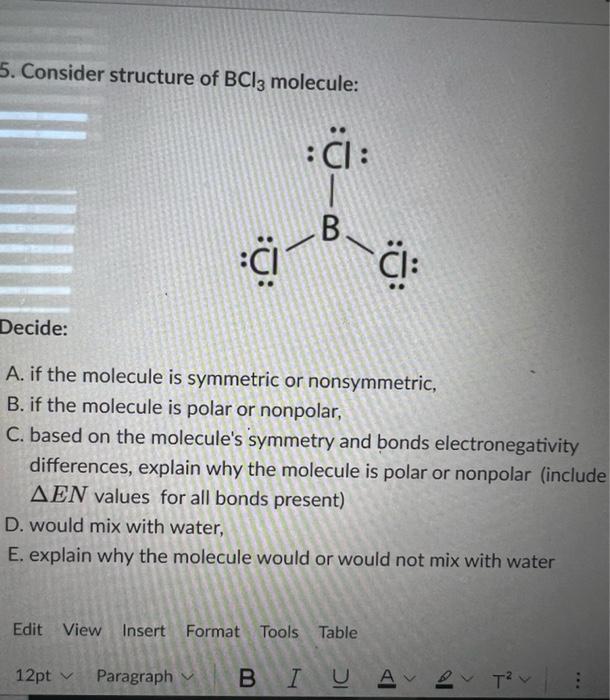
Consider the following statements about BCl3 molecules. Which statement is false? A. The B-Cl bonds are quite polar. B. The bond dipoles exactly cancel. C. The Cl-B-Cl bond angles are 109.5 degrees.